The International Aluminum Institute (IAI) predicts the global demand for aluminum in the electronics industry will rise from 8 million tons to 12 million tons in the coming years. However, the demand for copper is expected to reach the same level (around 13 million tons). Which begs the question, just how good of a conductor is aluminum?
Understanding the electrical conductivity of aluminum is essential for designers, product developers, and companies who want to benefit from the cost-effective nature of aluminum or its lightweight properties.
In this blog, we’ll cover aluminum conductors, the electrical characteristics of aluminum, and their possible use as an alternative to copper and other conductive metals.

Aluminum as a Metal
Before we delve into the conductivity of aluminum and its electrical properties, here are some basic facts about aluminum.
This background knowledge will help you understand the properties table and make comparison between aluminum conductors easier.
Aluminum is a metal element in the periodic table with the atomic number 13. It is one of the most abundant metals found on earth. Unfortunately, aluminum does not exist in its pure form and has to be produced at an industrial scale from its ore–Bauxite.

Aluminum conductor is widely used in the electronics industry, and it is the 4th most conductive metal after silver, copper, and gold; in that order. Many alloys of aluminum have different conductivities.
Aluminum is lightweight, ductile, and corrosion-resistant, which helps it in electrical applications because it can be turned into wires and quickly melted for soldering.
So what’s the significance of the number 13? It represents the number of electrons in an atom of aluminum and their arrangement in electron orbitals. Later, we will find that it is these electrons that contribute towards aluminum’s conductivity.
How to Measure Conductivity?
Conductivity tells you how well a metal can conduct electricity. It is a measure of a material’s tendency to conduct electrical current.
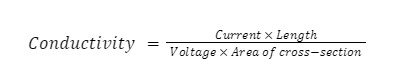
Conductivity is measured in Siemens (S) per meter (m). For aluminum, a known current is passed through the aluminum sample, and the voltage drop is measured using probes.
This can be used to calculate the conductivity of aluminum metal using a formula.
Can Aluminum Conduct Electricity?
Aluminum is one of the best electrical conductors. It is just behind pure copper in terms of conductivity. Pure aluminum conductor has an electrical conductivity of 33.3 x 107 S/m.

To easily understand conductivity, it is often compared to that of pure copper. Pure copper is the best electric conductor behind silver. Which, for economic reasons, is not viable to use as a conductor at an industrial scale.
The International Annealed Copper Standard (IACS) gives a comparative reference for conductivity. Aluminum is 61% IACS, meaning it has an electric conductivity that measures 61% to that of copper.
Aluminum is widely used in electrical equipment because of its cost-effectiveness and low electrical resistance. Aluminum weighs about 30% lighter than copper, making it ideal for overhead power cables.
Aluminum is such a good conductor of electricity that transmission lines are mostly made of aluminum, and the electric current coming to your house comes via aluminum conductors.
How Does Aluminium Conduct Electricity?
If you’re a designer understanding how aluminum conducts electricity can help you decide between aluminum alloys and other metals for your project.
Aluminum can conduct electricity because of its structure and atomic bonding.
This section covers how aluminum can conduct electricity.
Background About Current
At the most basic level, you’d think current is a measure of electricity. That’s not entirely true. Current is the rate at which the charge passes through a point inside a loop.
To understand how aluminum conductors work, you need to understand ‘charge,’ which is a positive or negative ion or electron.
Aluminum’s Conduction
Aluminum atoms are made of protons and neutrons, tightly held in the core, and surrounded by electrons.
When an aluminum atom loses an electron, it is called an aluminum ion–a positively charged aluminum ion.

Aluminum is made of many of these atoms, which are tightly packed together and have their outer electron as free electrons. They are not held tightly by the core of the aluminum atoms and can move around the aluminum.
To be specific, aluminum atoms have 13 electrons and 13 protons. This is why it is marked 13 on the periodic table. Arranging these 13 electrons in orbitals results in 3 electrons in its outer shell, which experience weak attraction from the nucleus.
Aluminum and other metals are surrounded by a sea of electrons that can move freely. Whenever there’s a potential difference across the ends of an aluminum piece, these conduction electrons can move in the direction of polarity (positive or negative).
This movement of free electrons makes aluminum conduct electricity, and a measure of the rate at which these electrons pass through a point is current.
How Does Aluminum’s Conductivity Compare To Other Metals
The conductivity test on aluminum can also identify the correct grades of aluminum and determine if a heat treatment process has been done on aluminum. A heat-treated aluminum sample has a different conductivity than untreated aluminum.
There are defined International Annealed Copper Standard ratios for different alloys, and treatment methods.
There’s conductivity variation from aluminum alloy to aluminum alloy and between aluminum and metals. This table from Researchgate compares the conductivity of aluminum with known metals.
| Rank | Metal | Electrical conductivity (Sm-1) |
| 1 | Silver | 66.7 106 |
| 2 | Copper | 64.1 106 |
| 3 | Gold | 49.0 106 |
| 4 | Aluminum | 40.8 106 |
| 5 | Rhodium | 23.3 106 |
| 6 | Zinc | 18.2 106 |
| 6 | Nickel | 16.4 106 |
| 7 | Cadmium | 14.7 106 |
| 8 | Iron | 11.2 106 |
From the table, you can quickly tell Iron is not the top choice for electrical applications. Iron is mainly used in its stainless steel form and is one of the worst conductors.
But why is copper ranked so high in terms of conducting electricity?
Why Copper is more Conductive than Aluminum?
For many reasons, copper is still the primary choice for electrical conductors. It is more ductile, has a golden-brown finish that can help aesthetics, and has better strength and hardness. Yes, it is an excellent conductor of electricity and better than aluminum.
Copper is a better electrical conductor than aluminum because of its resistance to electric current.
Resistance is a measure of opposition to current flow. Materials with high resistance, like plastic or rubber, are called insulators. All materials have resistance, but in metals, it is extremely small.
Resistance depends on dimensions and a material property called resistivity. If you consider copper conductors with equal length and area as their aluminum counterparts, you’ll find copper conductors to have high conductivity. This is because copper has a smaller resistivity than aluminum.

In experimental terms, copper is a good conductor because its free electrons are less likely to experience a phenomenon called phonon collision. This is when vibrating atoms produce a mechanical form of energy that interferes with electron movement. As a result, aluminum’s free electrons, despite being in a higher concentration than copper, experience more phonon collisions and scatter about, or in other words, contribute to resistance to electric current.
Aluminum Grades and Electrical Conductivity
In its raw form, aluminum has a low resistance to electric current. However, that’s not how aluminum is used. Most applications of aluminum require a specific treatment, addition of impurity, or formation of alloys.
Aluminum alloys are classified in grades. Aluminum grades start from 1000 series and range to 8000 series. You can read more about aluminum grades and their best applications here.

1000 series aluminum is the purest and most conductive. 2000 series are alloys of copper, and 6000 series are magnesium-silicon and aluminum alloys.
There’s no steadfast rule for guessing the conductivity of aluminum grades. Instead most data is experimental.
EC-grades of aluminum conduct electricity best. This is a 1000 series aluminum (1350 Al) with good electrical and thermal conductivity. EC refers to electrical grade. It has approximately 61 % IACS conductivity.
Other grades of aluminum like AA-8006 and AA-8011 are also good conductors with applications including aluminum wire and construction.
Aluminum’s Finish and Conductivity
Aluminum’s surface finish can change its electrical properties. Simply put, if you use aluminum for electronic devices and end up with some form of surface alteration, it can diminish aluminum’s conductivity. Hence, defeating your original purpose of having a good conductor.

Aluminum has a natural aluminum oxide layer that is extremely thin. Many people want their products to last longer and perform anodizing which can increase its corrosion resistance by growing the oxide layer.
However, the aluminum oxide layer does not conduct electricity because it has no free electrons. This way, you’re surrounding aluminum with an extremely poor conductor of electricity.
Powder coating, heat treatment, paint, and plastic coatings all affect aluminum conductors differently. As mentioned, their conductivity ratio to IACS is sometimes used to identify which heat treatment has been done on aluminum.
For example 6009-T4 is 44 % IACS, while 6009-T6 is 47% IACS. T4 and T6 are different types of heat treatment processes for the same aluminum alloy.
Applications of Aluminum in Electronics and Electrical Products
There are many reasons to use aluminum as an electrical conductor. However, its properties make it ideal for a few particular purposes in the electrical industry.
Wiring and Cabling
Aluminum wire is often used in overhead transmission lines because aluminum has a low density than copper. This means it is lighter in weight than copper.
Using aluminum wire over copper wire results in three times lighter weight, which helps prevent these cables from sagging. So, for a 1 m aluminum line weighing around 6 Kg, an equivalent copper conductor of 18 Kg would be required.

Aluminum offers around 60% – 64% IACS, which is an excellent conductivity compared to its weight and abundance.
Aluminum is also more cost-effective to produce and easier to handle than copper.
While houses and construction sites use copper wires more, industries and significant power generation sources employ aluminum wires. The 8000 series aluminum wires have excellent conductivity and are used in electrical wiring of residential buildings.
Electrical components
Aluminum’s electrical conductivity is also used to make electrical components from small connectors to large electronic devices where its lightweight properties can be used.

A rapidly growing market for aluminum is electric cars, where the casings for the motor and the electric motor use aluminum to keep the heavy battery from adding more weight.
Heatsinks
Its thermal conductivity makes it ideal for heatsinks and is often found in computers and processing units. They are also used in lamps and amplifiers.

Aluminum heatsinks essentially increase the area of heat dissipation which results in rapid cooling and quicker heat transfer away from the electronic device.
Conclusion
Aluminum is one of the best conductors with ever-expanding uses. It has a light weight, low cost, and ready availability worldwide, making it ideal for many electrical uses.
Aluminum is one of the two commercially used conductors besides copper. Despite being slightly less conductive than copper, its lightweight makes it extremely useful in many industries, from automobiles to power grids.
The conductivity of aluminum is so high that a simple aluminum foil can act as an electrical conductor. However, the conductivity of the foil will be much less than that of a wire or pure aluminum.
This is because the surface features of aluminum affect its conductivity. Painting, coating, or anodizing can significantly decrease conductivity. So, you should consider the final application of your aluminum product before considering any surface treatments.





